
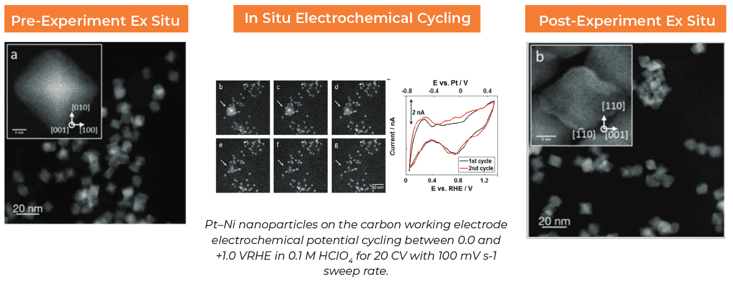
Observing Catalyst Stability in the TEM, A Workflow
Written by Nynke Krans, Applications and Marketing Manager, Apr. 26, 2024
An Arsenal of Tools for the Preparation of In Situ Electron Microscopy Samples
Written by Nynke Krans, Applications and Marketing Manager, Apr. 15, 2024

The New and Improved Success Community
Written by Madeline Dukes, Senior Applications Scientist and Training Specialist, Apr. 10, 2024

Gas Analysis in Atmosphere AX Using an RGA
Written by Tim Eldred, Product Marketing Manager, Mar. 20, 2024
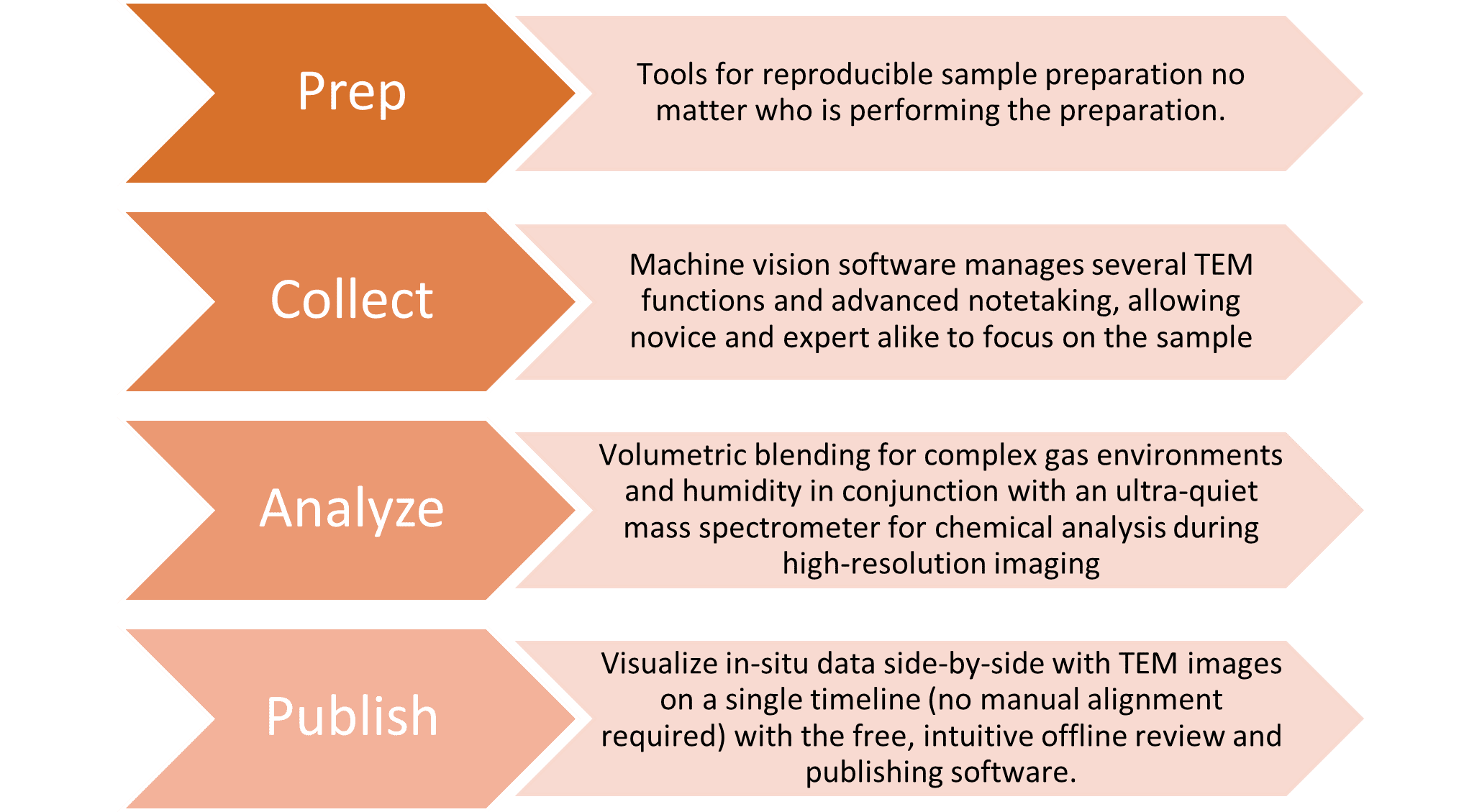
Workflows for Gas-Phase Heterogeneous Catalysis Research
Written by Jennifer McConnell, Senior Manager, Marketing and Product Management, Mar. 10, 2024

Flow Management E-chips for Electrochemistry
Written by Jennifer McConnell, Senior Manager, Marketing and Product Management, Jan. 10, 2024
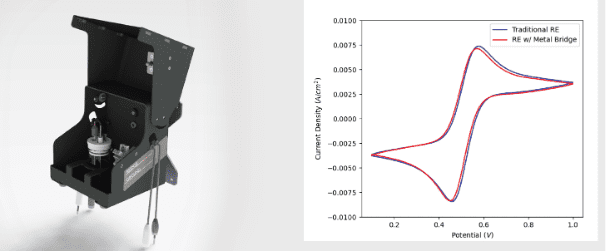
Using a Standard Reference Electrode during In Situ Electron Microscopy
Written by Nynke Krans, Application Specialist, Oct. 15, 2023

Protochips Launches In Situ AX
Machine Vision Powered Solutions for your TEM Applications, Apr. 13, 2023

