Machine Vision Liquid Cell for TEM
Generate relevant in situ TEM results that complement the bulk scale, opening a direct portal to understanding the causes of material changes in their native environment and at the nanoscale. Using the Poseidon AX, relevant and realistic environments can be added to your nanoscale research studies, such as battery studies, nanoparticle synthesis or life science.
We offer a complete solution from sample preparation to reporting results. Poseidon AX will push performance, productivity, and discovery to exponentially high levels for your lab. With the incorporation of first-of-its-kind machine-vision software technology (AXON Synchronicity) the capacity and productivity of the microscopist is enhanced. Combining the AXON software with our in situ liquid cell, the capture of high resolution and high value data with fewer iterations, and streamlines large data set processing and reporting is enabled, both inside and outside of the TEM lab.
System components
Learn more about what the system is comprised off
Click here to see all components of the systemIn situ liquid cell research examples
with the Poseidon AX system
with the Poseidon AX system
Biological Materials
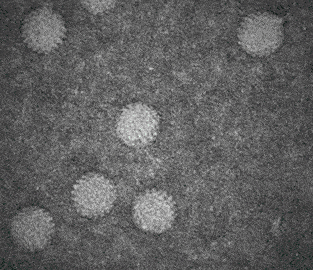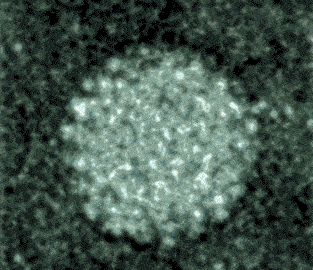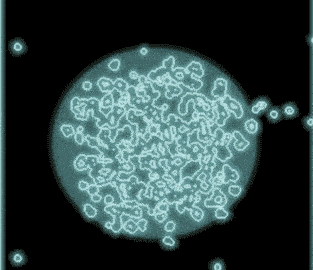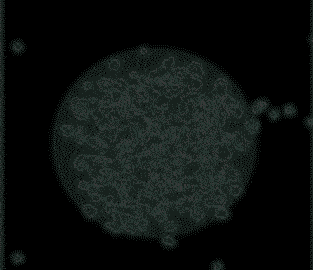
Observing virusses, polymers, lipids and other life-science samples at the nanoscale is possible using the Poseidon AX system. In this case the mobility of rotavirus particles was observes using our specialized microwell E-chips.
Varano, A.C. et al. (2015) Chem. Com. (51) 16176–16179
Click here to download an overview on biological materials using Poseidon AX

Biomineralization
The Poseidon AX system comes with 2 inlet ports to allow optimal mixing at the tip. This clip shows the formation of calcite using a protein mediate growth by mixing.
Perovic, I. et al. (2014) Biochem. (53) 7259–7268.
Click here to download an overview on biological materials using Poseidon AX

Nanoparticle synthesis
The Poseidon AX system can be used to heat liquids to a maximum temperature of 100° C. In this experiment the growth of gold nanoparticles was observed under different temperatures, creating a variety of nanoparticle shapes and sizes with high control.
Khelfa, A. et al. (2021) J. Vis. Exp. (168) 62225
Click here to download an overview on nanoparticle synthesis using Poseidon AX

Electrocatalysis
Using the Poseidon AX system, an electrochemical bias can be applied in a three electrode set-up to any system. In this case, a CuSO4 solution was observed growing dendrites using cyclic voltammetry.
Click here to download an overview on electrocatalysts using Poseidon AX

Batteries
Most in-depth understanding of plating and stripping behavior in batteries and the mechanisms by which adverse dendritic growth may occur remains underdeveloped. With Poseidon AX, these plating mechanisms can be investigated at the nanoscale.
Pu, S.D. et al. (2020) ACS Energy Letters, 5, 2283–2290
Click here to download an overview on batteries using Poseidon AX
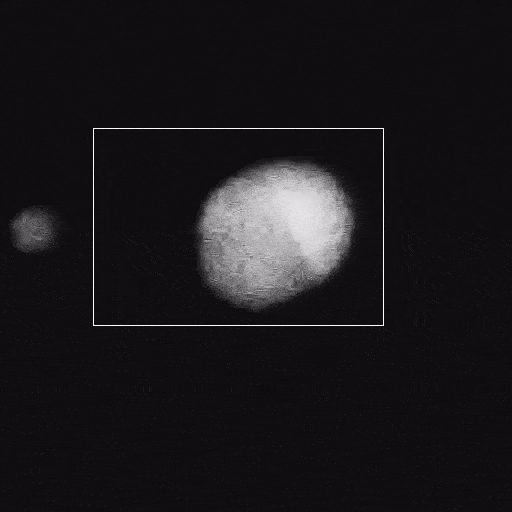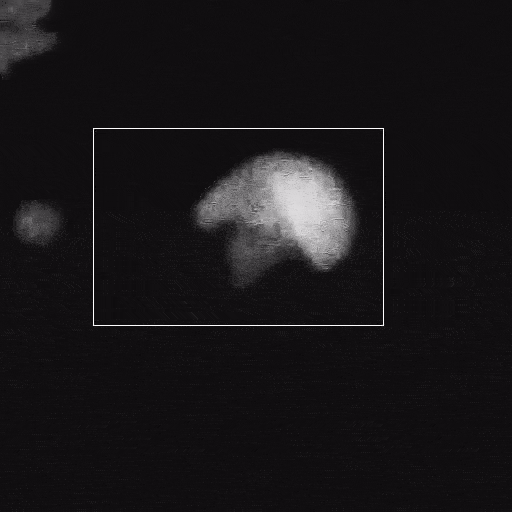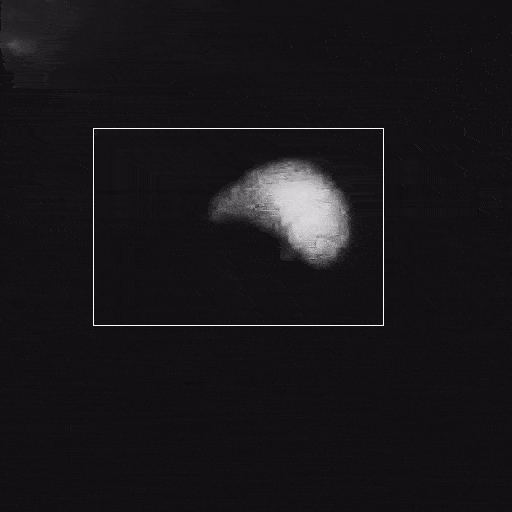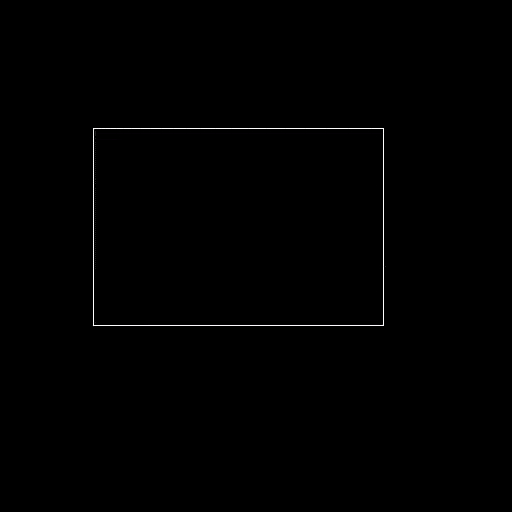
Corrosion
Corrosion is a significant problem for the stability of structural metals and potentially for functional nanomaterials in operating environments. The Poseidon AX system can be used to study corrosion of nanoparticles or FIB lamella.
Du, J.S. et al. (2021) Adv. Funct. Mater. (31) 2105866
Click here to download an overview on corrosion using Poseidon AX

Catalysis Synthesis
Catalysts work directly on the nanoscale to convert reactants into products. The synthesis of these catalysts needs to be controlled to make sure the materials have a good activity, selectivity and stability. In this case a Fischer-Tropsch catalyst is being synthesized in real time by adding iron oxide colloids in liquid to carbon nanotube supports.
Krans, N.A. et al. (2019) Micron, (117) 40–46
Click here to download an overview on nanoparticle synthesis using Poseidon AX
TEM Processing Software: AXON Studio
AXON Studio is an intuitive processing software for managing, reviewing, analyzing and publishing TEM images and metadata in your office or workspace. The software can be downloaded free of charge and operates by linking the TEM and in situ data to your images. AXON Studio can be used to plot, filter and publish videos, images and raw data. The AXON Studio software works hand-in-hand with the AXON Synchronicity TEM software which is linked to the microscope. This together forms the AXON platform.

